Contact process is widely adopted for production of sulfuric acid which is the world’s most consumed chemical, and its production relies on a highly specialized process known as the Contact Process. At the heart of this operation is the catalytic conversion of sulfur dioxide SO2 to sulfur trioxide SO3, a step governed by fascinating, yet challenging, thermodynamic principles. Understanding the Contact Process is crucial for optimizing every aspect of a modern sulfuric acid plant. The Contact Process ensures efficiency and effectiveness in sulfuric acid production. The contact process is indispensable for maximizing production efficacy.
In the context of industrial production, the Contact Process plays a pivotal role in achieving high yields and quality.
Understanding the SO2 to SO3 is not just academic—it is the foundation for optimizing every aspect of a modern sulfuric acid plant, particularly through the Contact Process. The significance of the contact process cannot be understated as it drives innovation in production techniques.
1. The Fundamental Challenge: Temperature, Conversion, and Equilibrium
The key chemical reaction, SO2 to SO3, is a classic exothermic reversible reaction, meaning it releases heat. The thermodynamics of this reaction are clearly summarized by the Typical Equilibrium Curve:
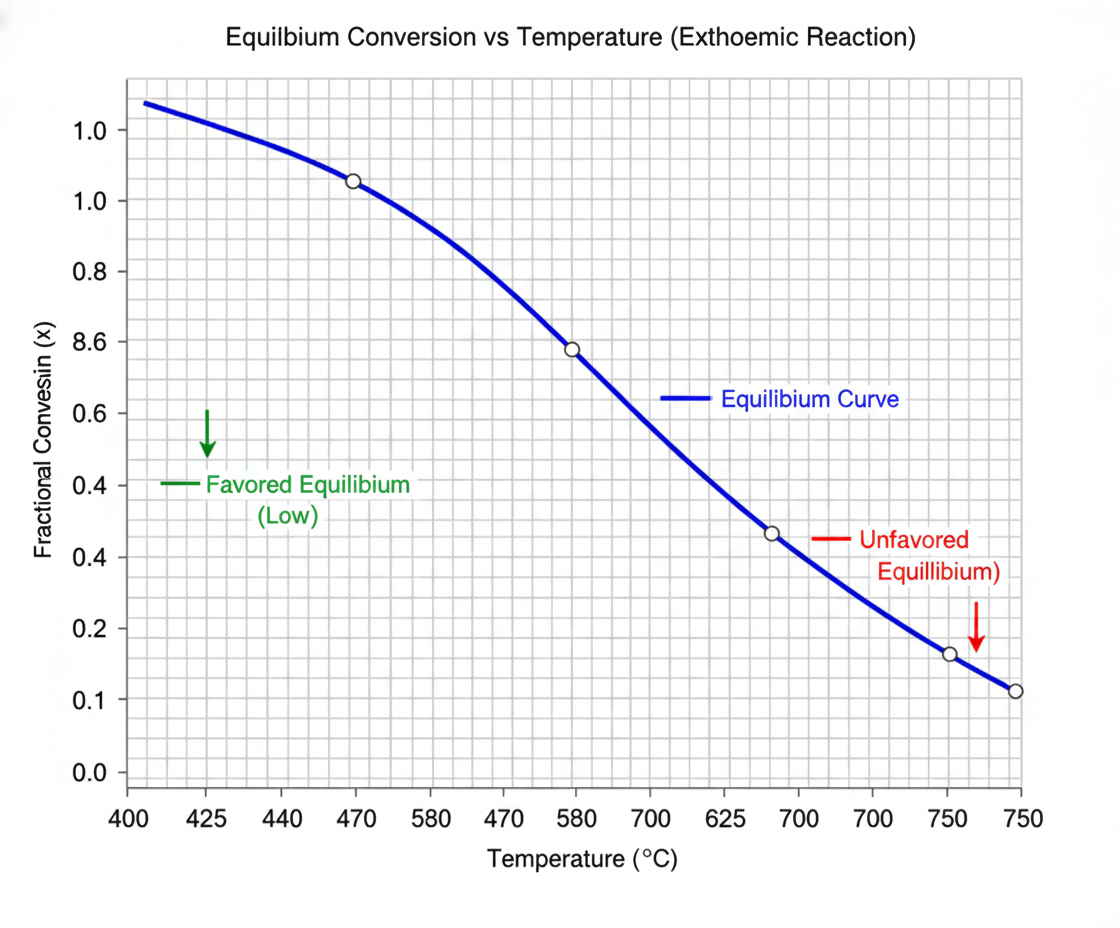
- Low Temperature is Favorable: At temperatures between 400 ℃ and 450 ℃, the equilibrium conversion rate can reach an impressive 95%–99%.
- High Temperature is Detrimental: As the temperature increases (e.g., up to 700 ℃–750 ℃, the equilibrium conversion rate plunges to 30%–40%.
This inverse relationship is the defining rule: Temperature rise causes the equilibrium to shift back towards the reactants SO2, limiting the maximum conversion achievable. This simple curve is the fundamental reason behind every decision made regarding cooling and temperature control in the Contact Process.
2. Adiabatic vs. Isothermal: Why a Multi-Bed Reactor is Essential
While the low-temperature conditions are ideal for maximum SO2 conversion, industrial reactors must also consider reaction kinetics (rate). We need the reaction to be fast enough to be economical.
The Industrial Reality: The Adiabatic Path
The industrial standard is the adiabatic reaction path. In an adiabatic system, no heat is removed, allowing the temperature to rise naturally as the reaction proceeds.
- As the exothermic reaction occurs, the temperature inside the reactor bed rises rapidly.
- This rise causes the reaction to follow an “upward sloping” path on the equilibrium curve.
- The system quickly approaches the equilibrium limit corresponding to that higher temperature, at which point the conversion rate effectively stalls.
The Solution: Multi-Bed Design and Inter-Bed Cooling (Contact Process)

If the reaction were run in a single, large adiabatic bed, conversion would quickly cap out at a low value. Therefore, the Contact Process uses a multi-bed adiabatic reactor design—a dynamic compromise between the speed needed for kinetics and the low temperature needed for high equilibrium conversion.
The basic logic is:
- Bed 1: Reactants enter and rapidly heat up (strong kinetics). Conversion is achieved until the system approaches the high-temperature equilibrium line.
- Inter-Bed Heat Exchange: The hot gas is removed and cooled (often via a heat exchanger).
- Bed 2, 3, 4: The cooled gas re-enters the next bed at a lower temperature. This shifts the equilibrium forward again, allowing for additional, high-efficiency conversion.
3. Optimizing Efficiency: Gas Strength and Temperature Control
The success of the multi-bed adiabatic reactor depends heavily on managing the reaction rate and temperature rise, especially concerning Gas Strength SO2 concentration).
- High SO2 Concentration (Gas Strength): Leads to a more vigorous reaction, releasing more heat. The adiabatic reaction line is steeper, meaning the temperature rises faster and the conversion is limited sooner.
- Low SO2 Concentration: The reaction releases less heat, resulting in a flatter temperature rise.
This control is critical for processes like Autotherm plants, where the heat generated in the first bed must be sufficient to “self-heat” the gas stream to the required ignition temperature of the subsequent beds.
Conclusion
The operation of a sulfuric acid plant is a masterful exercise in balancing thermodynamics and kinetics. The entire process hinges on respecting the limitations set by the SO2 to SO3 Equilibrium Curve. By breaking the conversion into discrete, adiabatic steps separated by controlled cooling (inter-bed heat exchange), plants are able to continuously reset the equilibrium, pushing overall conversion rates past 99% and ensuring efficient, high-yield sulfuric acid production.
You can continue to read the other posts by click here




